CelltrionHealthcare
Eydenzelt™
-

- Product Name : Eydenzelt™
- INN : Aflibercept
- Indications :
Treatment of neovascular (wet) age-related macular degeneration (nAMD), diabetic macular edema (DME), myopic choroidal neovascularization (myopic CNV), and macular edema following retinal vein occlusion (RVO).
- Protein Type : Recombinant fusion protein
- Drug Approval Status : Health Canada approved
-
Remsima™ SC is the world's first subcutaneous formulation of biosimilar infliximab developed by Celltrion. It was approved by Health Canada for rheumatoid arthritis on January 28, 2021, and for Crohn’s disease and ulcerative colitis on February 15, 2024.
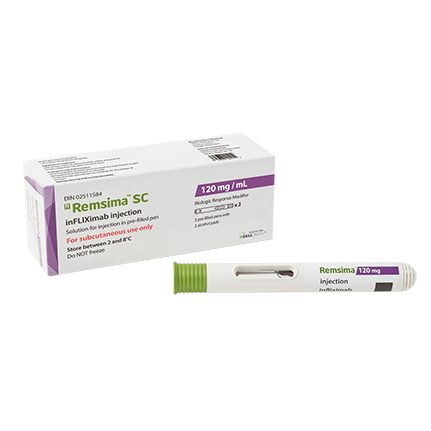
- Product Name : Remsima™ SC
- INN : Infliximab
- Indications : rheumatoid arthritis Crohn’s disease ulcerative colitis
- Protein Type : Monoclonal antibody (mAb)
- Mechanism of Action : It slows down the disease progression by neutralizing tumor necrosis factor-alpha (TNF-a), which is a common cause of autoimmune diseases.
- Drug Approval Status : Health Canada approved
*This product description is for medical and educational purposes only. They are not intended to promote PR or sales campaigns.
*For more details, please consult a doctor or a medical specialist.
*This site is intended for all over the world. -
Yuflyma® is a low volume/high concentration biosimilar of adalimumab available in 40 and 80mg strengths in both Auto injector and pre-filled syringe formats. It was approved by Health Canada on December 24, 2021.
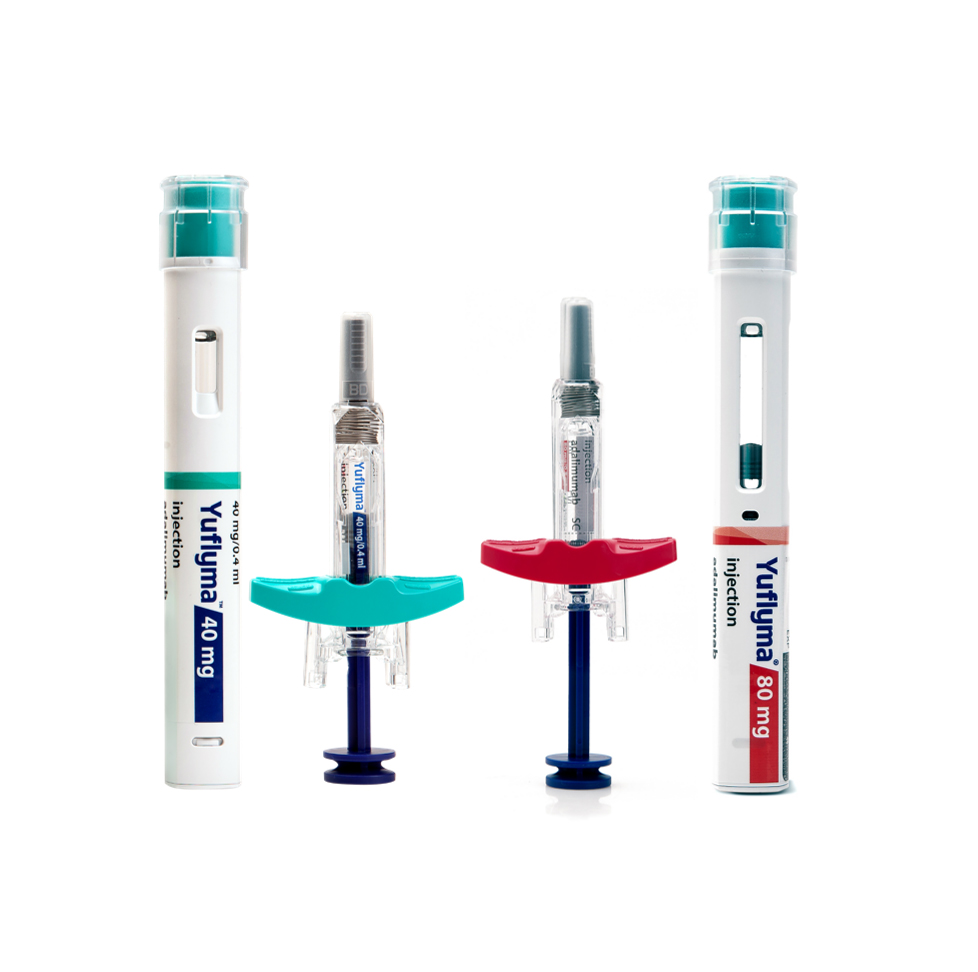
- Product Name : Yuflyma™
- INN : Adalimumab
- Indications : Rheumatoid arthritis Psoriatic arthritis Ankylosing spondylitis Psoriasis Adult Crohn’s disease Adult ulcerative colitis Polyarticular juvenile idiopathic arthritis Adult and adolescent hidradenitis suppurativa Adult and pediatric uveitis
- Protein Type : Monoclonal antibody (mAb)
- Strength : 40mg/0.4mL, 80mg/0.8mL
- Drug Approval Status : Health Canada approved
*This product description is for medical and educational purposes only. They are not intended to promote PR or sales campaigns.
*For more details, please consult a doctor or a medical specialist.
*This site is intended for all over the world. -
Vegzelma™ is a biosimilar of bevacizumab developed by Celltrion. It was approved by Health Canada on January 3rd, 2023.
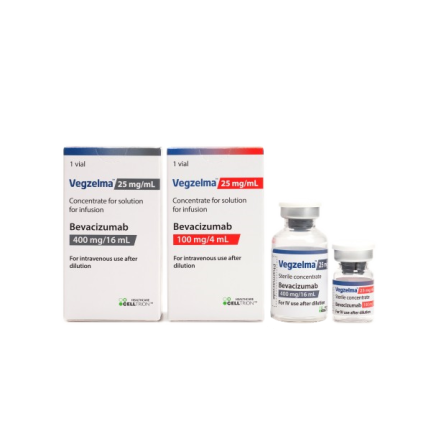
- Product Name : Vegzelma™
- INN : bevacizumab
- Indications : Metastatic Colorectal Cancer (mCRC) Locally Advanced, Metastatic or Recurrent Non-Small Cell Lung Cancer (NSCLC) Platinum-Sensitive Recurrent Epithelial Ovarian, Fallopian Tube and Primary Peritoneal Cancer Platinum-Resistant Recurrent Epithelial Ovarian, Fallopian Tube and Primary Peritoneal Cancer Malignant Glioma (WHO Grade IV) – Glioblastoma
- Protein Type : Monoclonal antibody (mAb)
- Strength : 100mg/4mL, 400mg/16mL
- Drug Approval Status : Health Canada approved
*This product description is for medical and educational purposes only. They are not intended to promote PR or sales campaigns.
*For more details, please consult a doctor or a medical specialist. -
Steqeyma® is a biosimilar of ustekinumab developed by Celltrion. It was approved by Health Canada on July 30th, 2024.
- Product Name : Steqeyma®
- INN : ustekinumab
- Indications :
- Plaque Psoriasis - Psoriatic Arthritis - Crohn’s Disease
- Protein Type : Monoclonal antibody (mAb)
- Strength :
45mg/0.5mL, 90mg/1mL (SC) 130mg/26ml (IV) - Drug Approval Status : Health Canada approved
*This product description is for medical and educational purposes only. They are not intended to promote PR or sales campaigns.
*For more details, please consult a doctor or a medical specialist. -
Omlyclo™ is the world’s first and only omalizumab biosimilar developed by Celltrion. It was approved by Health Canada on December 6th, 2024.
- Product Name : Omlyclo®
- INN : Omalizumab
- Indications :
Allergic Asthma (adult and pediatric patients – 6 years of age and above) Chronic Rhinosinusitis with Nasal Polyposis (CRSwNP) Chronic Idiopathic Urticaria (CIU)
- Protein Type : Monoclonal antibody (mAb)
- Strength :
IgE plays a central role in the pathophysiology of inflammatory diseases in the airway. Omalizumab binds to IgE and prevents binding of IgE to the high-affinity IgE Receptor, FcεRI, thereby reducing the amount of free IgE that is available to trigger the allergic-inflammatory cascade. - Drug Approval Status : Health Canada approved on December 6th, 2024
*This product description is for medical and educational purposes only. They are not intended to promote PR or sales campaigns.
*For more details, please consult a doctor or a medical specialist. -
- Product Name : Remdantry™
- INN : infliximab
- Indications :
Rheumatoid arthritis Ankylosing spondylitis Crohn’s disease (adult and pediatric) Fistulising Crohn’s disease Ulcerative colitis (adult and pediatric) Psoriatic arthritis Plaque psoriasis
- Protein Type : Monoclonal antibody (mAb)
- Strength :
100mg/mL - Drug Approval Status : Health Canada approved on Jan 15th, 2014* Health Canada approval on brand name change on Mar 10th,2025
*Previously distributed under the tradename Inflectra®. Inflectra® is a registered trademark of Pfizer Inc.
*The product image may not accurately represent the version available in Canada. -
- Product Name : Stoboclo™
- INN : Denosumab
- Indications :
Is approved for treating osteoporosis in postmenopausal women and men at high risk for fracture, for increasing bone mass in men with non-metastatic prostate cancer on androgen deprivation therapy and women with non-metastatic breast cancer on adjuvant aromatase inhibitor therapy who are at high risk for fracture, and for increasing bone mass in women and men at high risk for fracture starting or undergoing long-term systemic glucocorticoid therapy.
- Protein Type : Fully human monoclonal antibody (IgG2)
- Drug Approval Status : Health Canada approved
-
- Product Name : Osenvelt™
- INN : Denosumab
- Indications :
Is indicated for reducing the risk of skeletal-related events in patients with multiple myeloma and in patients with bone metastases from solid tumours, the treatment of adults and skeletally mature adolescents with giant cell tumour of bone, and the treatment of patients with hypercalcemia of malignancyrefractory to intravenous bisphosphonates.
- Protein Type : Fully human monoclonal antibody (IgG2)
- Drug Approval Status : Health Canada approved
-
- Product Name : Avtozma™
- INN : Tocilizumab
- Indications :
Treatment of moderate to severe rheumatoid arthritis (RA), active polyarticular juvenile idiopathic arthritis (pJIA), active systemic juvenile idiopathic arthritis (sJIA), CAR-T cell-induced severe or life-threatening cytokine release syndrome (CRS), and Coronavirus disease 2019 (COVID-19).
- Protein Type : Humanized monoclonal antibody (IgG1)
- Drug Approval Status : Health Canada approved